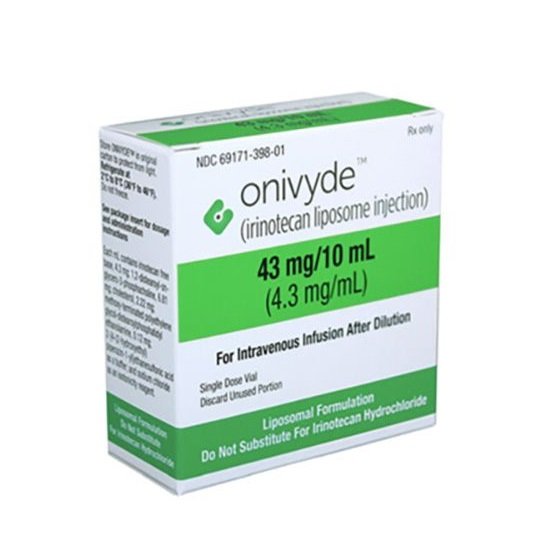
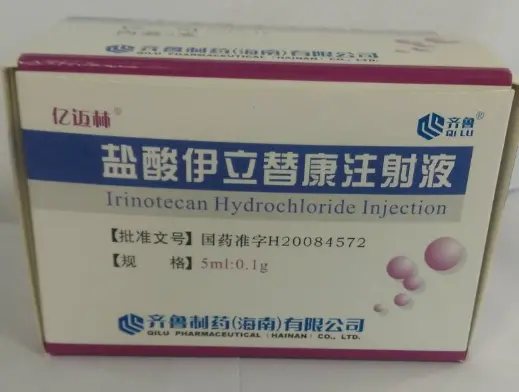
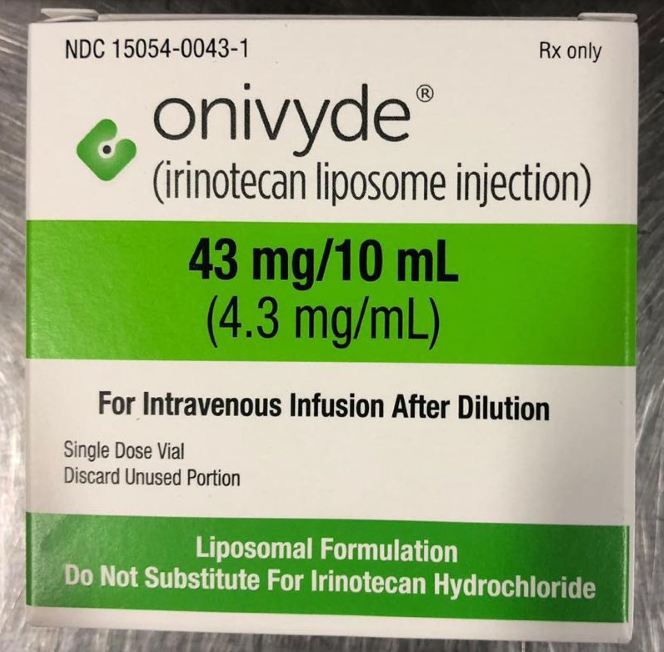
Irinotecan Hydrochloride Injection.
Efficacy:
Irinotecan hydrochloride is suitable for the treatment of patients with advanced colorectal cancer: combined with 5-fluorouracil and folinic acid to treat patients with advanced colorectal cancer who have not received chemotherapy before. As a single drug, it is used to treat patients who have failed chemotherapy regimens containing 5-fluorouracil.
Usage and Dosage:
Recommended dose In monotherapy (for patients who have received previous treatment), the recommended dose of this product is 350 mg/m, intravenous infusion for 30-90 minutes, once every 3 weeks. In combination therapy (for patients who have not received previous treatment), the safety and efficacy of combined use with 5-fluorouracil (5-FU) and folinic acid (FA) were evaluated by the following regimen: a 2-week treatment regimen of this product plus 5-fluorouracil (5-FU)/folinic acid (FA); the recommended treatment dose of this product is 180 mg/m, administered once every 2 weeks, continuous intravenous infusion for 30 to 90 minutes, followed by infusion of folinic acid and 5-fluorouracil. Dose adjustment should be performed after all adverse reactions have returned to grade 0 or 1 of the NCI-CTC (National Cancer Institute Common Toxicity Criteria) grading standard and treatment-related diarrhea has completely resolved. At the start of the next infusion treatment, the dose of this product and 5-fluorouracil (if this drug is used) should be reduced according to the most severe adverse reaction observed in the previous treatment. In order to facilitate the recovery of treatment-related adverse reactions, treatment should be postponed for 1-2 weeks. If the patient still cannot recover after a delay of 2 weeks, chemotherapy should be stopped. When the following adverse reactions occur, the dose of this product and/or 5-fluorouracil (if this drug is used) should be reduced by 15-20%. Hematological toxicity [neutropenia grade 4, febrile neutropenia (neutropenia grade 3-4, fever grade 2-4), thrombocytopenia and leukopenia (grade 4)] Non-hematological toxicity (grade 3-4) This drug should be used continuously until objective lesion progression or unbearable toxicity occurs. Special populations Monotherapy for patients with impaired liver function: In patients with high bilirubin and clotting time greater than 50%, irinotecan clearance is reduced, and thus its blood toxicity increases. Complete blood counts should be performed frequently in this population. When the patient’s bilirubin is within 1.5 times the upper limit of normal (ULN), the recommended dose of this product is 350mg/m2; when the patient’s bilirubin is 1.5-3 times the upper limit of normal, the recommended dose of this product is 200mg/m2; when the patient’s bilirubin exceeds 3 times the upper limit of normal, this product cannot be used for treatment. Combination therapy: There is currently no data on the use of patients with impaired liver function. Patients with impaired renal function: No studies have been conducted on patients with renal insufficiency.
Drug contraindications:
Allergy to this product is contraindicated. Contraindicated during pregnancy. Contraindicated during lactation. Use with caution in patients with impaired liver and kidney function
Related dosage forms:
Injection, injection
Share:
Products
Our offers
Health Classification
Let us work together to protect precious health