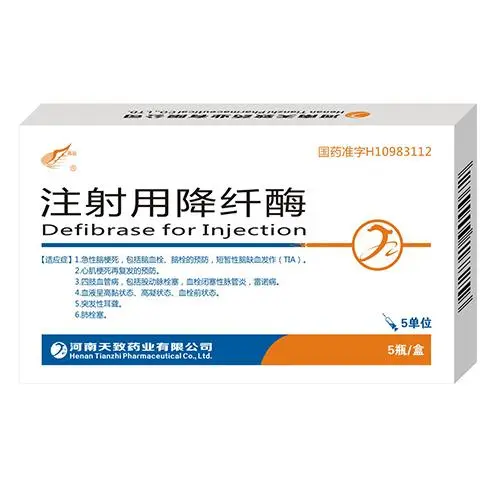
Difibrase, DF
Function:
For the prevention of acute ischemic stroke, including cerebral thrombosis, cerebral embolism, transient ischemic attack (TIA), and recurrence of cerebral infarction; for the prevention of myocardial infarction, unstable angina, and recurrence of myocardial infarction; It is used for vascular diseases of the extremities, including femoral artery embolism, thromboangiitis obliterans, and Raynaud’s syndrome; it is used when blood is in a hyperviscous state, hypercoagulable state, and prothrombotic state; it is used for sudden deafness; it is used for pulmonary embolism.
Dosage:
Intravenous drip. Acute attack period: 10U once a day, for 3-4 days. Non-acute attack period: 10U for the first time, 5-10U for maintenance, once a day or every other day, 2 weeks as a course of treatment. Defibrase powder injection (i.e. defibrase for injection) is first dissolved in water for injection or 0.9% sodium chloride injection, and then diluted with 100-250ml of 0.9% sodium chloride injection; defibrase injection is in 100-250ml Use after dilution with 0.9% sodium chloride injection. When taking medication, pay attention to controlling the intravenous infusion speed, and the infusion time should be more than 1 hour. If the infusion rate is too fast, uncomfortable symptoms such as chest pain and palpitations may occur. Activities should be reduced within 5-10 days after taking the medication to prevent bleeding caused by accidental trauma. In addition, patients should have fibrinogen and other bleeding and coagulation function tests before and during medication, and pay close attention to clinical symptoms.
Drug contraindications:
If you are allergic to this product, use with caution during pregnancy. Use with caution in children. Use with caution during lactation.
Related dosage forms:
Injection, powder for injection
Share:
Products
Our offers
Health Classification
Let us work together to protect precious health